#titration
Text

Not my meme, but we did titration in chem today and this channels my every thought.
67 notes
·
View notes
Text
What is Acid Base Titration?
Acid base titration in chemistry is an experimental procedure used to calculate the unknown concentration of an acid or base by an indicator.

Many inorganic and organic compounds can be determined in the laboratory by an acid base titration based on their acidic or basic properties more about Acid Base Titration
12 notes
·
View notes
Text
Day three. Let's see what two is like
2 notes
·
View notes
Text
For the titration curve in figure 11.17, the initial pH will be:
pH = -log[H3O+]
= -log(0.200)
= 0.70

"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quote#chemistry#nonfiction#textbook#logarithmic curve#logarithm#titration#acidic#alkaline#sodium hydroxide#hydronium
2 notes
·
View notes
Text

5 notes
·
View notes
Text
(Recall from chapter 11 that the smaller the pKa the more acidic is the group. At lower pH, carboxylic acids are found in the RCOOH form and amines are found in the RNH3+ form. At higher pH, the opposite is true; carboxylic acids are present as the salt RCOO- and amines are present as uncharged RNH2. Figure 24.3 on p. 1061 shows how this looks at different pH.)
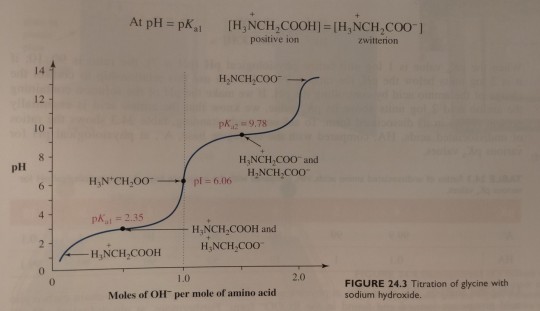
Next, the solution is titrated with 1.00 M NaOH; the volume of base added and the pH of the resulting solution are recorded and then plotted as shown in figure 24.3. (...) By examining the titration curve (figure 24.3), you can see that the isoelectric point for glycine falls halfway between the pKa values for the carboxyl groups and the ammonium ion:
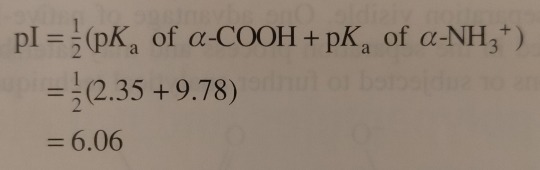
"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quotes#chemistry#nonfiction#textbook#acidic#carboxylic acid#ph#salt#amine#titration#glycine#sodium hydroxide#carboxyl#ammonium#ions
1 note
·
View note
Text

Day 2/14, 1st September, 2023 – Friday
Today wasn't that great a day either, I worked a little on my Chemistry Lab Report and emailed my classmates the values my friends and I had gotten in the lab prior, but otherwise it was pretty slow and uninspired. I'm trying to nudge myself out of my bad habits by posting for this challenge (can you tell haha), so hopefully that begins happening soon.
#studyblr#ib#ib studyblr#international baccalaureate#dp1#productivity challenge#study motivation#chemistry#lab report#titration
0 notes
Photo

#redox#titration#permanganate#oxalate#acidic#solution#manganous#salt#carbon dioxide#water#balanced#equation#coefficient#reactants#products#chemistry#solutions
1 note
·
View note
Text
The last week has been interesting with the titrating of my antidepressants, tonight, I titrate down to one....and next week I'm on no antidepressants whatsoever.
I'll be like this the day after the second part of an assessment for my DID....the first part triggered me and let the little one out which for some reason made things really bad ever since.
Yeah, I'm sure there will no problems with this in any way shape or form....
0 notes
Text

just one more reanimator shitpost
#reanimator#re-animator#danbert#herbert west#daniel cain#herbert's titration game is surely sick tho
3K notes
·
View notes
Text
me, when the dude and chick across from me decided to flirt for the whole lab period

#like???#titrations are horrible enough why they gotta make that shit unbearable 😭😭😭#smfh at all these titrations and shit#*horror narration* but then along came the STRAIGHTS#😱#it’s also made worse by the fact that he is the most Dude Bro#but apparently he just got voted frat prez good for him 🙌#i hate chemistry and it hates me back#RIP#analytical chemistry#chem lab#i hate titrations#titration#science meme
1 note
·
View note
Photo

【 土曜日 NLP murmur 】 「地圖」不是「實際狀態」, 而他確實可以幫助你認清方向⋯⋯ 當你我在創傷狀態時, 有些時候, 會以為自已在一個困局, 這就是妳的創傷地圖。 殊不知,在這不舒服的地圖上, 這些症狀也是讓我們走出來的鑰匙。 你可以去覺知自己身上的症狀感受, 用下面的九個步驟,幫自己, 走出創傷自癒的路: 1. 為你自己創造相對安全的環境 2. 允許自己探索與接納身體。 3. 善用生命節奏擺動/停止。 4. 利用滴定原理,一點點去鬆動,改變大腦與身體的認知。 5. 用主動防禦取代被動受創,做好你的界線管理。 6. 區分「恐懼」與「無助」。 7. 慢慢有次序去釋放/分配全身的求生能量 8. 回到動態平衡,自我歸零調整 9. 適應環境,覺知當下。 祝福妳能允許自己, 好好享受週末時光。 #創傷 #自癒 #地圖不等於實地 #認清方向 #症狀 #平衡 #動態平衡 #滴定 #symptoms #map #balance #titration #fear #helpless #freed #release #dynamicequilibrium #samwgnlp #nlpcat #samwgco https://www.instagram.com/p/CgDvyYFpJOb/?igshid=NGJjMDIxMWI=
#創傷#自癒#地圖不等於實地#認清方向#症狀#平衡#動態平衡#滴定#symptoms#map#balance#titration#fear#helpless#freed#release#dynamicequilibrium#samwgnlp#nlpcat#samwgco
0 notes
Text
idk if 3 is too many or if my kid was just extra annoying yesterday. Gonna try staggering 2 and see if that helps
0 notes
Text
At this point, the concentration of the zwitterion equals that of the positively charged ion, and the pH of 2.35 equals the pKa value of the carboxyl group (pKa1):
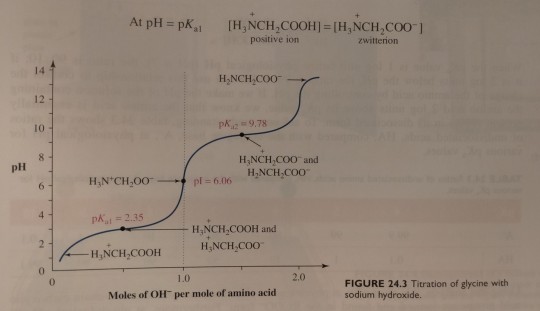
"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quote#chemistry#nonfiction#textbook#concentration#zwitterion#ions#carboxyl#titration#glycine#sodium hydroxide#chemical reactions
0 notes
Text
Phenolphthalein, for example, changes a solution from colourless to pink as the pH of the solution changes over a range of 8.2 to 10.0 (see table 11.9).

"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quotes#chemistry#nonfiction#textbook#acid#base#indicator#color change#titration#methyl green#thymol blue#methyl orange#ethyl red#methyl purple#bromocresol purple#bromothymol blue#phenol red#litmus#cresol red#phenolphthalein#thymolphthalein#alizarin yellow r#clayton yellow
0 notes
Text

cant stop looking at this image and crying with laughter. the boots. the matching poses. the matching purple cravats. whats the DEAL
#pers#kh#org13#all three of you need to pull your hair back for lab safety#exept ansem i know for a fact isnt in a lab doing anything he just like. wanders around#even and ienzo know what a titration is and have proper pipetting procedure + know how to work in a hood#ansem found a random ass machine and stuck a 7 year old in it to see what would happen. BE FOR REAL
105 notes
·
View notes