#kingdraw
Video
heres my ava6 episode 2 prediction
#kingdraw#mine#video#ava#alan becker#animator vs animation#i spent 30 minutes tops on this#and if you found the youtube mirror no you didn't
642 notes
·
View notes
Text
The chemistry behind Roasted Sweet Potatoes!

Choosing a perfect winter snack? Roasted sweet potatoes 🍠win hands down!
Hot, sweet, and irresistible, they owe their enticing aroma to the Maillard reaction during high-temperature baking. Key contributors include 2-Acetylfuran and 2-acetylpyrrole, delivering almond, caramel, and bakery notes.


#winter#sweet potatoes#roasted#chemistry#organicchemistry#cold weather#kingdraw#chemblr#stemblr#molecule#science#did u know
34 notes
·
View notes
Photo

bird man
#bnha#mha#boku no hero academia#my hero academia#hawks#bnha spoilers#takami keigo#art#kingdraws#500#1k
12K notes
·
View notes
Text

Drew this at the height of the out of touch meme and then didn’t post it, so here you go
#art#kingdraws#digital art#artists on tumblr#chainsaw man#chainsaw#chainsaw man art#angel devil#angel devil fanart#chainsaw man angel fanart#chainsaw man angel devil#akiangel#implied akiangel
16 notes
·
View notes
Photo

lets try another thing
#furry#Furry Fandom#Furry Art#fursona#digital art#digital drawing#jack skellington#canine#wolf#artists on tumblr#kingdraws#WolfInDeersClothing
4 notes
·
View notes
Photo

Day 9 - #pencilkings @100daysofsketchingofficial @xppen_india #100daysofsketching #100days2020 #100dayswithxppen #dooodlebuzz #pencilkingsdrawingchallenge #lionKing #keshrt #silhouette #lionkingmokey #kingdrawing https://www.instagram.com/p/B_-S8iOlqAc/?igshid=i9afvmhxeupe
#pencilkings#100daysofsketching#100days2020#100dayswithxppen#dooodlebuzz#pencilkingsdrawingchallenge#lionking#keshrt#silhouette#lionkingmokey#kingdrawing
0 notes
Photo

This be a birthday present for @king-draws
Have a good day hun
64 notes
·
View notes
Note
DUDE I just discovered you from that Danny Phantom animation meme you posted on YouTube and it blew me away, and your art is stunning too! Keep up the great work!
TYSM dude!!! The DP animation took some time so im super excited that you like it!
Dude but like?? have you seen your own art???? ITS so so flattering to get a compliment from someone who has as good an art style as you i reaLLY LOVE YOUR WORK
#HOLY SHIT DUDE AAAA#THANK YOU#ITS COOL WHEN TALENTED PEOPLE THING IM TALENTED#BRO YOUR ART IS JUST SO NICE FML#IMMA FOLOW YOU NOW TBH YOUR ART IS S O#ILUAGWUDHJSBX#jay speaks#EVEN though the entire meme is just tweening lmao#people are hard to draw#THANKS FOR THE KIND WORDS MY DUDE#x-kingdraws-x
8 notes
·
View notes
Text



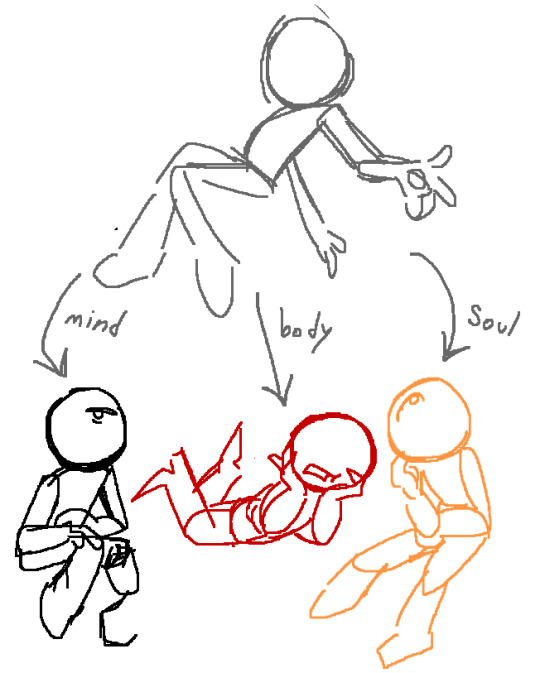
icks
#kingdraw#mine#ava#alan becker#animator vs animation#ava the second coming#ava green#ava victim#ava the chosen one#ava the dark lord#i can't explain my thought process for that last image you'll just ahve to trust me
125 notes
·
View notes
Text
Meet 'Pepper X,' the New World's Hottest Pepper

In August, the Guinness World Records crowned 'Pepper X' the world's hottest pepper, officially scoring 2.69 million SHU on the spiciness scale. That's hotter than pepper spray! For reference, the everyday jalapeño hits around 30,000 SHU. Kudos to Ed Currie for a decade of work in creating this fiery marvel, surpassing his own previous 'Carolina Reaper' at 1.64 million SHU. Currie, along with only four others, has dared to eat a whole Pepper X due to its extreme heat. The fiery sensation in peppers mainly comes from a group of closely related compounds known as 'capsaicinoids,' with capsaicin (69%) and dihydrocapsaicin (22%) leading the way.


#guinness world records#pepper#pepper x#spicy food#chemistry#molecule#organicchemistry#chemblr#kingdraw#stem studyblr#amazing facts#did u know#interesting#pls share
52 notes
·
View notes
Text

miku challenges you to a duel
581 notes
·
View notes
Text

I did some art fancy that!!
#art#kingdraws#digital art#artists on tumblr#chainsaw man#chainsaw man angel devil#angel devil#chainsaw man angel fanart#chainsaw man art#fanart#angel devil fanart
3 notes
·
View notes
Photo



these guys
#i dont post art to main much anymore so we'll see what happens#ava#alan becker#animator vs animation#kingdraw#ava the chosen one#ava the dark lord#ava victim
222 notes
·
View notes
Text
The chemistry behind Diya oil lamps

Diwali, India's traditional festival of light, sees the illumination of Diya oil lamps symbolizing prosperity and happiness.
In the darkness of this celebration, the burning candles and oil lamps may contain eugenol and ocimene from spices like basil, infusing the festival with delightful aromas. Eugenol and ocimene are natural compounds, commonly found in essential oils of basil, lavender, and artemisia, renowned for their aromatic qualities. Used in culinary and cultural activities, they add flavor to traditions. Interestingly, ocimene can be reduced to dihydromyrcenol in the presence of sodium and alcohol, while heating can cause isomerization to allo-ocimene.
KingDraw extends its warm wishes for a healthy and joyous year to all celebrating Diwali!


#diwali#chemistry#science#kingdraw#molecule#organicchemistry#dailychem#oil lamp#eugenol#ocimene#chemblr
43 notes
·
View notes
Text
Name Reaction, The Julia olefination

The Julia olefination (also known as the Julia–Lythgoe olefination) is the chemical reaction used in organic chemistry of phenyl sulfones with aldehydes (or ketones ) to give alkenes (olefins) after alcohol functionalization and reductive elimination using sodium amalgam or SmI 2. The reaction is named after the French chemist Marc Julia.

#chemistry#organicchemistry#chemical reactions#science#kingdraw#dailychem#chemblr#did u know#uni studyblr
36 notes
·
View notes
Text
A molecule that has the acronym DEAD!
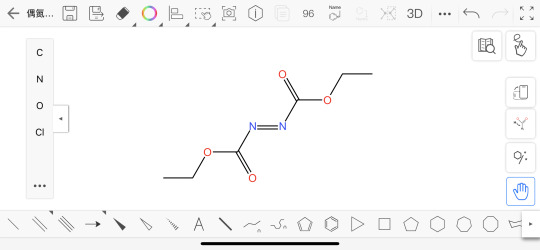
It stands for diethyl azodicarboxylate, an orange–red liquid that becomes yellow when diluted in a solvent. DEAD is a strong electron acceptor; it oxidizes iodide to molecular iodine, hydrazine to molecular nitrogen, alcohols to aldehydes, and thiols to disulfides. DEAD’s principal use is as a reagent in the Mitsunobu condensation reaction, and it participates in Michael and Diels–Alder reactions. Because it explodes when heated, it cannot be shipped in pure form. Instead, it is transported in solution or adsorbed onto plastic particles.

#stem studyblr#chemistry notes#chemblr#stemblr#chem student#molecule#organicchemistry#chemistry#kingdraw
51 notes
·
View notes