Text
My sexual orientation:

65 notes
·
View notes
Text
























These are some TF2 References that I've made! I've also posted them on Twitter If y'all are interested! I hope this helps you draw the mercs a bit easier, I know it's definitely helped me out!!!
6K notes
·
View notes
Text
When sodium hypochlorite (bleach) solution is added to luminol, a chemical reaction occurs that releases energy in the form of light. This is called chemiluminescence. The bleach solution acts as an oxidizing agent, which means it takes electrons away from the luminol molecule. This causes the luminol molecule to become excited, and it releases the energy as light.
🎥 Courtesy: Kendra Frederick
The luminol molecule is made up of two amino groups, a carbonyl group, and an azo group. The amino groups are electron-rich, while the carbonyl group is electron-poor. The azo group is a conjugated system, which means that the electrons in the double bonds can move freely from one atom to another.
When sodium hypochlorite (bleach) solution is added to luminol, the bleach molecules react with the amino groups of the luminol molecule. This reaction takes electrons away from the luminol molecule, which causes the luminol molecule to become oxidized. The oxidized luminol molecule is in an excited state, which means that it has more energy than it normally does.
The excited luminol molecule then releases the extra energy as light. This light is called chemiluminescence. The light emitted by the chemiluminescence reaction is blue because the luminol molecule has a blue fluorescence.
The chemiluminescence reaction between luminol and sodium hypochlorite is catalyzed by the presence of a metal ion, such as iron or copper. The metal ion helps to stabilize the excited state of the luminol molecule, which makes it more likely to release the extra energy as light.
The chemiluminescence reaction is very sensitive to impurities, so it is important to use pure chemicals. The reaction can also be affected by the pH of the solution. The optimal pH for the reaction is around 9.
The chemiluminescence reaction between luminol and sodium hypochlorite can be used to detect blood, as the iron in hemoglobin can catalyze the reaction. The reaction is also used in some commercial products, such as glow sticks and emergency lights.
I hope you enjoyed learning about this. ❤️🙏
5K notes
·
View notes
Text

#Barney looks like a typical CIS guy#my art#art#barney calhoun#hl2#hl2 barney#half life 2#hl2 fanart#half life fanart#digital art#artists on tumblr#artwork#half life#half life 2 fanart
825 notes
·
View notes
Text
I don’t understand why Half Life 2 fans underestimate Barney??? the dude literally suffered for 20 years after the cascade resonance☠️He survived the black mesa, zen, portal storms, 7-hour war, cover of city 17, took care of Eli, Kleiner and Alix, led the uprising (while Alix and Gordon were away for 2 weeks)
17 notes
·
View notes
Text

#my art#art#digital art#alyx vance#half life alyx#half life 2#half life fanart#half life#hl2 fanart#hl2#gordon freeman#hl2 gordon#freemance#artists on tumblr
234 notes
·
View notes
Photo

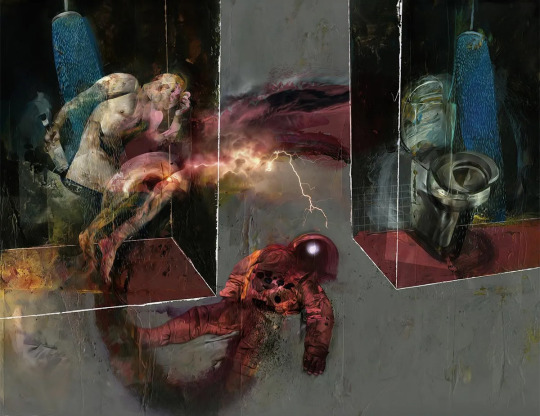
Dave McKean’s illustrations for Arkady & Boris Strugatsky‘s Roadside Picnic.
193 notes
·
View notes
Text


293 notes
·
View notes












