#Crystal Fuel
Text
26
*The glamour that had been growing blurrier and frailer by the minute breaks, melting off and dissolving*
*Glass underneath is much paler, blue tint to her skin. The place around her eyes seem stucken as well as her cheeks*
*She's shivering again*
*The Nameless worble, nervous whispers fill the air and she pushes away from Raphael and pushes herself up wheezing*
// @tinydancerandthemoonchild
25 notes
·
View notes
Text


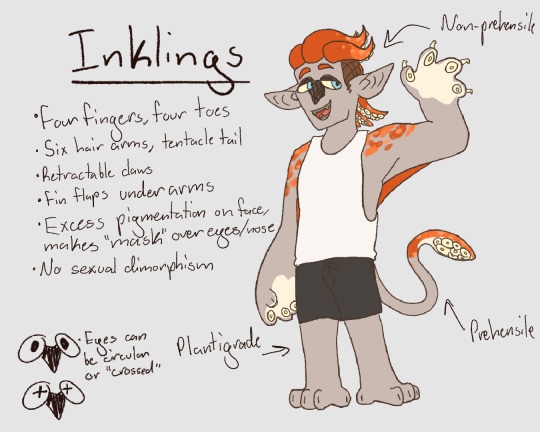
RAGH my own take on xeno designs
#thepossumpile#splatoon#xeno inkling#xeno octoling#splatvat#inspo taken from others designs real squids/octopuses and the game obviously#main goal wasn’t exactly realism#but rather a fun to draw design#also since we know canonically that alternas crystals fueled the evolution of the sapient species#i imagine mammal characteristics can sprout from that#which is where octolings get their nails#but inklings have their claws from the actual hooks squids have#please ask me xeno questions i think its fun to come up withstuff
143 notes
·
View notes
Text
IDK if I've posted this before but Ideal Location for Some Fireworks compilation
(Excluding all two locations where it might be reasonable to set off some fireworks)




#lotro#yeah go ahead why dont you#set off a firework directly behind a guard#also im in love with this last one i love it so much#my suspicion is that the location was there before the forge master and her stand was#but they forgot to move the location when they added her in#so now her stand gets blown up every festival#at least she has a good sense of humor about it#or at any rate ive never heard her say anything#maybe shes bottling it up and will eventually have enough and go on a rampage#a firework fueled rampage through the town#exploding anfalas star lit crystals and symbols of the elder king while shes at it
7 notes
·
View notes
Text
CRYSTALIZED EP 14 SPOILERS




he’s gonna bite you
no but oni lloyd is genuinely kinda scary
#ninjago#lloyd garmadon#oni lloyd#crystalized ep14#crystalized#crystalized part 2#low key nightmare fuel#that NOoOOo though#sam vincent is killing it#s15#s16#spoilers#crystalized spoilers
185 notes
·
View notes
Note
Do you have any headcanons on the effects of energon on human physiology? E.g. prolonged exposure (just being around it), skin contact, fumes, etc. I'm torn between the idea of making it like gasoline/petrol/bleach (with fumes and whatnot) vs making it more "safe", more like an oil (probably causes dryness/irritation/etc. in long term, but if you drop a bit on you, it won't kill you)
to me it depends on continuity/specifically what relation humans have to the Cybertronian mythos. like, in canons where Earth is Unicron, i believe energon as Primus's blood may actually be metaphysically toxic more than physically, but dark energon may have the opposite effect. in canons like TFA where none of that is relevant, i figure it probably depends on the physical state of the energon. crystal won't be nearly as harmful as liquid, etc
in general i like to think in liquid fuel form it can give people some pretty severe rashes or chemical burns when splashed or dunked, or even temperature burns if it's highly charged enough. a few droplets won't do much harm if brushed off quick though. i also think energon as a "perfect/near-perfect" fuel source actually doesn't produce much if any fumes, even when burned by an engine, so it's probably fine for humans to be around it if safely contained? maybe processing the crystal to liquid creates fumes/aerosolized bits that would be hell to breathe but, apart from being explosively reactive, i don't typically think of energon as being harmful to exist around. long term incremental exposure danger, sure, but with some good OSHA compliance you should be fine
#inspired more by like mercury and mineral poisoning and stuff rather than earth fuels#i always think of it in crystal form before i think of it as a fuel#considering im literally looking at my toxic rock collection rn there is a LOT of inspo to be had about toxicity in mineralogy!#energon#cybertronian food#transformers
15 notes
·
View notes
Text
Okay y'all I'm being that mutual. To those of us watching tscs tomorrow right when it drops, what's the spoiler tag? tscs spoilers? Bc like. I need to block this shit so I don't get mad at work lmao
I'll be tagging with tscs spoilers and also dani watches tscs so that if my yelling gets annoying you can block it and keep enjoying 😁😁
#I will save the angies for the evening and fuel crystal springs motivation with it!!!!!#text#tscs#tscs spoilers#I'm very negative i know i know#LEAVE ME TO IT!#and remember to tag your spoilers 😁#dani speaks#the santa clauses
8 notes
·
View notes
Text
rotating in my head the way the bureaucracy of the twin adders hates eyrie for weird legal situations
#it’s a funny legality of how after the bloody banquet eyrie was discharged from the twin adders#1) to protect the adders from any conflicts with the crystal braves and 2)#to keep eyrie safe by keeping either side in ignorance as to the affairs going on#they were reinstated after the Dragonsong war but there was a catch—eyrie refused to accept it#legally they have not accepted being reinstated as an officer for the twin adders#that is fueled not by spite over the banquet but because they never wanted to join in the first place#it was pressed upon them by the scions that they join a grand company#and eyrie was never really keen to be tied down—an attitude they wanted to protect as they grew in fame#as a member of the scions they didn’t want to be politically attached#and they certainly would be politically attached#plus being back in a military setting after their time with the garleans was NOT the move#a military hierarchy is the same no matter what—politics included#the way it makes them feel to be around that environment again is. not great#but yeah they refuse to accept it and live in a limbo state#they very easily could be discharged again but they don’t like waving power around like that#and they do get some amusement out of being a bother#even if they are just a small one#owen talks#oc: eyrie kisne
5 notes
·
View notes
Text
input jerma into an ai so that it would create a realistic screen grab from an 80’s dark fantasy movie and created possibly the hottest unreal movie villain of all time




#jerma985#jerma#ai#ai generated#I AM NOT ONE OF THOSE AI GENERATED ART GUYS BTW. i just love creating images to fuel my dumb ideas#ai art is bad and dumb but the dark crystal jerma is not
23 notes
·
View notes
Text
for my discord rp oomfs that follow me its funny how im seen as the bitch crazy over rp ships when im ngl im lowkey more insane over the crystals that heighten your mental illness
#ingrambles#no cuz. the crystals dont create the problems#they HEIGHTEN them#they fuel the paranoia of a person's pre existing worried and fears#smth smth the crystals being the embodiment of trauma#they make me go insane girl
6 notes
·
View notes
Note
the uhhhh fairy god from windwaker that looks creepy and controls the fairyts like puppets but Zelda
oubbff... i always thought she looked like fi which is also making me think of sword spirit zelda. which i am of course very normal about too ,,
#i think generally i just want zelda to be weird and creepy and fucked up. i want her to terrify me and i want the whole goddess thing to be#explored in a horror fueled way. i wnt her to be scary so soo bad ok shes been put into so many crystals she deserves it i think
5 notes
·
View notes
Text
29
*She's silent*
*The Nameless chatter* - Glass
// @tinydancerandthemoonchild
25 notes
·
View notes
Photo

#it#pennywise#clown#creepy clown#scary clown#kirby#kirby 64#kirby 64 the crystal shards#ripple star queen#queen ripple#creepy#memes#half and half#nightmare fuel
11 notes
·
View notes
Photo

I can’t find the episode to this but here’s Tom Baker’s doctor winking at you for your pleasure.
#wink!#those teeth#WHAT IN THE NAME OF SANITY#DOES THE DOCTOR BRUSH HIS TEETH WITH?!#don't mention those piercing eyes#fueling my heart with true excitement#could look at fourth doctor all day#the doctor#doctor who#fourth doctor#tom baker#smile#grin#hello!#eyes#teeth#sphere#crystal ball#mirror#gif#emote#winking#WONK
5 notes
·
View notes
Text
Vivzie should give us the new episode as an Easter present. Just saying.
#im feral bro#i need fuel for stolas angst cus you know blitz is taking that crystal and fucking off
1 note
·
View note
Text
Molecular Weaving Makes Polymer Composites Stronger Without Compromising Function - Technology Org
New Post has been published on https://thedigitalinsider.com/molecular-weaving-makes-polymer-composites-stronger-without-compromising-function-technology-org/
Molecular Weaving Makes Polymer Composites Stronger Without Compromising Function - Technology Org
At its most basic, chemistry is a lot like working with building blocks at its most basic level, but the materials are atoms and molecules. COFs – or covalent organic frameworks, a new class of porous crystals – are a great example of a material that behaves like a molecular Lego set, where individual building blocks are connected through strong chemical bonds to form a highly open and structured network.
This intricate structure provides a scaffold for polymer chains to thread or wrap during their formation and strength. Think of a woven scarf or basket – a single piece of yarn or twine may not be much on its own, but when woven together, the pattern enhances the final product’s overall performance. Furthermore, when these chains weave together, sometimes even the chemical reactions further strengthen the material’s properties.
Schematic illustration of the COF structure, polymers, and nanofibrils courtesy of Science Magazine / UC Berkeley
In 2016, Yaghi Research Group, led by UC Berkeley’s Professor of Chemistry Omar Yaghi, realized the first molecularly woven structure by interlacing the backbone of the framework in a 3D space. These molecular woven COF crystals are tough but extremely flexible, as every atom has a high degree of freedom to move around but is also locked in place, and as a whole the woven crystals are able to dissipate energy during stress to prevent fracture.
Today, together with Ting Xu, Professor of Chemistry and Materials Science & Engineering; and Rob Ritchie, Professor of Materials Science & Engineering, the lab is now leveraging both the porosity and molecular weaving to make polymer composites stronger, tougher, and more resistant to fracture by threading polymer strands through the woven network. Their findings have been published in a paper by��Science.
“This is exciting because most filler materials enhance one mechanical property at the detriment of another,” said Ephraim Neumann, a PhD candidate at the College of Chemistry working at the Yaghi Research Group. Neumann is sharing his first authorship with joint student of Xu and Ritchie, Junpyo (Patrick) Kwon, who graduated (PhD) last year from UC Berkeley.
But why are COFs themselves so useful to everyday life? One example is that due to their exceptional porosity, COFs are used extensively in storing and separating gases such as hydrogen and methane. Both hydrogen and methane are clean energy carriers that can be used in fuel cells and combustion engines. Storing them enables their use in transportation and power generation without producing harmful emissions.
Now, thanks to this new research that suggests polymer composites can be made more durable, the applications and uses have wider implications.
“When we add a small amount (1%) of these woven COF crystals to other materials such as polymer or plastic in this case, the materials become significantly tougher and can have a high tolerance for damages and fractures. This could have a huge impact on the materials industry,” said Yaghi.
For example, polyimide, found in almost every laptop and electrical wiring, was one of the investigated polymers in this study. By adding woven COF nanocrystals, the team was able to improve the mechanical performance of the polymer without compromising its thermal stability. This suggests this technique could lead to longer lifetimes for these composites. “Or if the material becomes more resilient, one could use less of it to achieve the same result,” hypothesized Neumann. Polyimide can also be found in the solar sails used by NASA, as it is often used as a support material that lends thermal and mechanical durability to many applications.
“Many properties of plastic products rely on polymer chain entanglements,” said Xu. “My favorite analogy is how an angel hair pasta and a bowtie pasta may respond to a swirl in the plate. Adding nanoparticles of these crystalline COFs can template how these long chains may arrange spatially and get the whole plate to work together. It also becomes feasible to pull out the chains, separate out polymers from COF nanoparticles and do the process again from scratch.”
When thinking about how this might affect industries beyond materials, Neumann concluded, “While this discovery focuses on specific polymers, the basic concept of using porous, molecularly woven COFs to enhance mechanical properties could be extended to many other materials.”
Source: UC Berkeley
You can offer your link to a page which is relevant to the topic of this post.
#3d#amp#applications#atom#atoms#Building#Cells#chemical#chemical bonds#chemical reactions#chemistry#clean energy#college#composites#crystalline#crystals#Emissions#energy#engineering#engines#form#framework#fuel#fuel cells#Fundamental physics news#how#hydrogen#illustration#impact#Industries
0 notes
Text

all the Mira posting just to share possibly the funniest out of context back and forth I wrote in a fever dream last night
#Mira looking at a little yellow crystal: my dad died and turned into crystal so I took a piece of it with me so he could see the universe#Mira: that is my biohazard and he has behaved impeccably return my father to me#I was bullshitting names of things so her previous employer has a stand in name (Vengeer Watch) and so does her planet#or more of a system name the locals probably called it something else (Saiven Alliance Planet 9-3)#I just wanted to make her picking her last name more fun to me (Sapphe comes from the planet being called SA P-93 for short)#I did name the crystal fuel Divenum but I don't Love that so it's more of a placeholder until I think of something better or cooler
1 note
·
View note