Photo



Kintsugi: the Japanese art of repairing broken pottery with gold-infused lacquer. The practice adheres to the philosophy of treating breakage and repair as natural parts of an object’s history, rather than incidents whose evidence is to be concealed (x)
Repaired Heart, Kintsugi Study #4) and Kintsugi Study #2 by TJ Volonis
36K notes
·
View notes
Video
LAST WARNING MOFO... THIS SHIT IS GONNA GET SERIOUS BRO!
722K notes
·
View notes
Photo


Kill the boy, Jon Snow. Winter is almost upon us. Kill the boy and let the man be born…
5K notes
·
View notes
Text
Nephrotic Syndrome
Mnemonic: Protein LEAC (LEAK):
Proteinuria
Lipid up
Edema
Albumin down (losing protein)
Cholesterol up
Note: In Nephrotic Syndrome, the proteins leak out.
© MedXclusive.Tumblr.com
246 notes
·
View notes
Photo

Wolf with ornaments by Daniel Meyer via LEITBILD
www.dasleitbild.com
www.leitbild.tumblr.com
www.facebook.com/dasleitbild
www.instagram.com/dasleitbild
www.pinterest.com/leitbild
345 notes
·
View notes
Photo

Clock & Blue Rose
http://tattooideas247.com/clock-rose-chest/
45 notes
·
View notes
Text
Valvular Heart Disease
Buckle your seatbelts kids. I’ve been doing some relearning of valvular heart disease today and I wanted to share because I often need a refresher on this stuff. I’m going to start by assuming you’ve forgotten everything about the anatomy of the heart and it’s valves, mostly because I always need a reminder when I get to thinking about the heart.
Passage of blood through the heart:
Deoxygenated blood from the body (superior/inferior vena cava) and deoxygenated blood from the walls of the heart (coronary sinus) → right atrum → through the tricuspid valve → right ventrical → through the pulmonary valve → lungs to become oxygenated → pulmonary veins → left atrium → through the mitral valve → left ventricle → through the aortic valve → aorta → body AND coronary arteries
But you already knew all that.
Now lets jump into the fun stuff.
What on earth is valvular heart disease? Well friends, you may be surprised to learn, it’s a disease, of the valves, of the heart. Lots of diseases actually. There are two main ways messed up valves cause problems:
1) Stenosis: failure of the valve to open completely. This impedes blood flow
2) Insufficiency: failure of the valve to close completely. This allows reversed flow. (You can also call this regurgitation or incompetence. Same concept.)
You can either have isolated disease in which only one valve is affected, or combined disease where more than one valve is affected.
The 4 most common categories of acquired heart valve disease are pretty easy to remember. We will get more specific, but let’s start big picture. Valvular lesions tend to affect the left heart – aka the mitral and aortic valve. And we already know that stenosis and insufficiency are the big bad guys. Put those ideas together and you get the 4 most common forms of acquired heart valve disease:
1) Aortic stenosis - ex. calcific aortic stenosis
2) Aortic insufficiency
3) Mitral stenosis - ex. Rheumatoid Heart Disease
4) Mitral insufficiency - ex. Lupus endocarditis
So how do these bad things happen to nice people?

Calcific Aortic Stenosis
Fun fact: your heart beats 30-40 million times per year. That means 30-40 openings/closings of those valves. That’s a lot of mechanical stress on the hinge points. Imagine flipping a soda pop tab back and forth. What happens? It breaks. Fortunately our hearts are slightly better equipped to deal with this type of stress, but the cumulative wear and tear does take its toll. In addition to the mechanical stress, there is also chronic injury due to hyperlipidemia, HTN, inflammation, etc… (the same types of things implicated in atherosclerosis). All of this incites the degeneration of the aortic valves and deposition of calcium salts (just like you find in bone!).
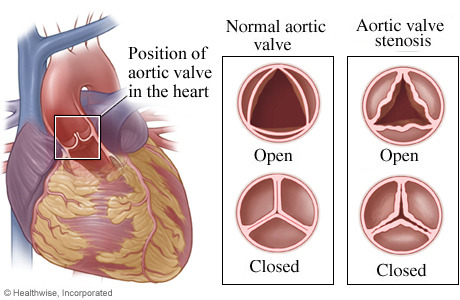
In individuals with normal aortic valve anatomy (three cusps) this damage tends to start causing clinical problems when people are in their 70’s-90’s (it’s called senile calcific aortic stenosis because it affects old people → that can’t be PC). Currently it affects about 2% of the population but that number is going up as people are living longer. In individuals with abnormal bicuspid aortic valves, clinical symptoms present 10-20 years earlier (in their 50s-70s) because bicuspid valves incur greater mechanical stress.
Clinical features: The calcific deposits on the aortic valve lead to poor opening and increased resistance to left ventricular outflow and increased left ventricular pressure. In order to compensate and get blood out into the body, the left ventricle has to work way harder, resulting in concentric left ventricular hypertrophy.

This big beefy heart isn’t as efficient, so it has to work hard AND it isn’t well perfused, because it’s too thick AND it’s squishing the coronary arteries (which probably also have atherosclerosis since it shares the same risk factors). This leads to angina symptoms. Without treatment, 50% of people who develop angina will die within 5 years. Eventually, the heart can’t keep up and you end up with cardiac decompensation. Blood backs up in the heart and you have congestive heart failure. 50% of people with CHF die within 2 years.

Mitral annular calcification
When there is calcification of the mitral valve, it tends to occur in the peripheral fibrous ring (annulus). This happens most often in women older than 60 and people with mitral valve prolapse. These deposits appear as hard ulcerated nodules at the bases of the leaflets. This can lead to stenosis or regurgitation depending on the exact location. It can also lead to arrhythmias & sudden cardiac death if the deposits get deep enough to interfere with AV nodal conduction. These nodules can also provide a site for thrombus formation (which increases risk or embolic stroke) or a nidus (a place in which bacteria have multiplied or may multiply) for infective endocarditis.
Mitral valve prolapse (myxomatous degeneration of the mitral valve)
In mitral valve prolapse (MVP) one or both of mitral valve leaflets are “floppy” and prolapse back into the left atrium during systole. (Remember, during systole the mitral valve should be closed so the ventricle can force blood out into the aorta, not back into the atria). Some cases are associated with Marfan’s syndrome, but there are plenty of cases without an obvious genetic connection. This condition affects ~3% of American adults, and typically it is an incidental finding, although there can be complications associated with it.
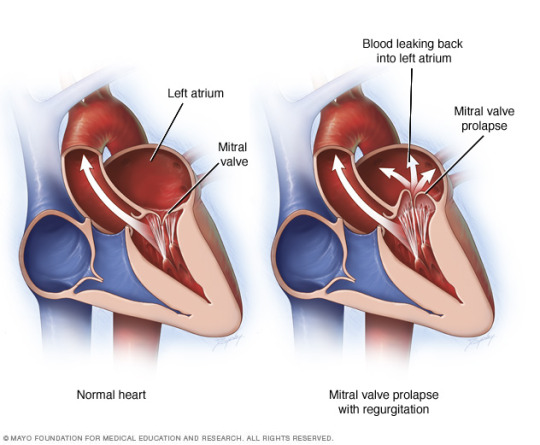
Clinical features: Most individuals with MVP are asymptomatic. The condition is revealed by a mid-systolic click upon auscultation and confirmed by echocardiography. Approximately 3% of people with MVP develop a complication such as infective endocardititis, mitral insufficiency (sometimes with chordal rupture), stroke or infarct due to embolism of leaflet thrombi, or arrhythmias (ventricular and atrial).
Rheumatic fever and heart disease
And you thought you were done with strep after that micro exam…

Rheumatic fever (RF) is a multisystem inflammatory disease classically occurring a few weeks (10 days to 6 weeks) after an episode of group A streptococcal pharyngitis. Antibodies and CD4+ T cells directed against streptococcal M proteins sometimes also recognize cardiac self-antigens leading to tissue damage. 1% of those affected die of heart failure and myocarditis. Diagnosis of acute RF is established using the Jone’s criteria. You need evidence of a group A strep infection (typically antibodies to streptolysin O & DNAse B) plus 2 of the following major signs: (thank First Aid for the mnemonic – J♥NES criteria)
- Joints → migratory polyarthritis of the large joints
- ♥ → pancarditis
- Nodules (subcutaneous)
- Erythema marginatum (a rash with clear margins typically on trunk and upper limb)
- Sydenham’s chorea (involuntary, purposeless movements)
Or one major sign and two minor signs (fever, arthralgia, elevated acute phase reactants, prolonged PR interval). Most cases (80%) are in people 5-15 years old. Carditis is more common in kids. Arthritis is more common in adults. In general, it it is less common to see RF in the US than in the developing world (because we usually treat strep throat before it progresses).
Pancarditis affects the whole heart.
1) Pericarditis: a fibrinous/serofibrinous exudate forms between the heart and the pericardium causing stickiness and rubbing (AKA Bread & Butter Pericarditis because it looks like when you butter two pieces of bread, stick them together and pull them apart). This usually resolves with time.

2) Myocarditis: features perivascular Aschoff bodies (foci of Tcells and activated macrophages called anitschkow cells).
3) Endocarditis: subendocardial inflammatory foci resulting from subendocardial lesions and regargant jets → may lead to formation of MacCallum plaques (irregular thickenings).
4) Valvulitis: inflammation of the valves, chordae tendinae, etc….
Inflammation of the endocardium and the left-sided valves typically results in fibrinoid necrosis within the cusps or tendinous cords. Small vegetations form over these necrotic foci and along the lines of closure.
In chronic RHD, you can see changes in the mitral valve including “fish mouth” or “buttonhole” stenosis and accompanying left atrial dilation.


In most cases (65-70%) of chronic RHD only the mitral valve is affected. In some cases the aortic valve is also involved, but RHD accounts for less than 10% of aortic stenosis.
Complications of CRHD include further damage (damage begets more damage), arrhythmias (ex. Afib with mitral stenosis), increased risk of thromboembolic events, and infective endocarditis.
Treatment involves surgical repair or prosthetic valve replacement
Infective endocarditis
Infective endocarditis is a microbial (typically bacterial) infection of the heart valves or endocardium. It results in vegetations, made up of organisms, inflammatory cells and thrombotic debris. Typically there is destruction of the underlying cardiac tissue.

^ not that kind of vegetation
Diagnosis is tricky because the signs and symptoms can be found in other illnesses, and blood cultures are not always positive (because those sneaky critters are hiding in the heart). Typically in cases suspected to be IE, you draw blood from multiple sites when the patient has a fever to increase the chances of getting something to culture. Still, in about 10-15% of cases no organism can be isolated (culture negative endocarditis).
The most important predisposing factor for IE is bacteremia, which can happen by any number of mechanisms (ex. dental/surgical procedures, contaminated needles, trivial epithelial barrier breaks). Interesting tidbit: You tend to see problems in the right heart in IV drug users because that’s where the veins dump in, and they are using dirty needles to shoot drugs into their veins. They also tend to be pick up nasty critters like staph aureus. Other critters that can cause IE include strep viridans (oral cavity flora), HACEK oral cavity commensals (Haemophilus, Actinobacillus, Cardiobacterium, Eikenella, & Kingella), staph epidermidis (prosthetic valves), Gram (-) rods and sometimes fungi.
Clinically, IE is divided into acute and subacute endocarditis. Acute endocarditis is typically caused by infection of previously healthy heart valves by highly virulent organisms (like staph aureus). You see destructive, friable vegetations which erode into underlying myocardium, lots of inflammatory cells, and multiple valve involvement. Patients present with rapid onset of symptoms and are difficult to cure. Subacute IE is caused by less virulent organisms that cause insidious infections of deformed or damaged valves. Patients present with vague, flu-like symptoms, but once identified, subacute IE can usually be resolved with antibiotics.
Clinical Features: Fever is the most consistent sign. Acute IE has rapidly developing fever, chills, weakness, and lassitude plus fatigue, weight loss and flu like symptoms. Complications include glomerulonephritis (due to Ag-Ab complexes). 90% of patients with left sided IE also have a heart murmur (either new or from an existing abnormality). In long-standing IE you may also see splinter or subungal hemorrages, Janeway lesions, Osler nodes, Roth spots (we talked about these guys in micro), intracranial hemorrhages, and/or abscesses/infarcts elsewhere in the body. You don’t typically see these later manifestations if you diagnose and treat early.
Diagnosis is based on the Duke criteria.
Noninfected vegetations/endocarditis
Noninfected vegetations occur in nonbacterial thrombotic endocarditis and Libman-Sacks endocarditis.
Nonbacterial thrombotic endocarditis (NBTE) is characterized by deposition of small, sterile, thrombi loosely attached to the leaflets of the cardiac valves along the lines of closure. These vegetations are not invasive so there is no local inflammatory reaction, but they may release emboli and product infarcts in the brain, heart, etc… (so they are still bad). Patients with a hypercoaguable state (venous thrombosis, pulmonary embolus, cancers, especially mucin producing adenocarcinomas) are at an increased risk of NBTE, as are patients with a history of endocardial trauma (ex. indwelling catheters).
Endocarditis of systemic lupus erythematosus (Libman-Sacks endocarditis) is associated with small, sterile, pink vegetations of the mitral (more common) and aortic (sometimes) valves. They may be located on the undersurface of the valves, which is unique. These can lead to regurgitation.
Recap of Different Vegetations:

Vegetations in RHD tend to form along the edges of the (usually mitral) valve leaflets. Vegetations in IE are large and irregular and cover valve cusps. They may extend into the chordae. Vegetations in lupus may be located on the undersurface of the valves.

Whew. That’s all I’ve got for now.
854 notes
·
View notes
Photo

NO?
Let’s learn about a not-so-negative word.. Yes, I’m talking about nitric oxide, not no. (If you got whatever I meant :P )
Nitric oxide (NO) is formed from L-arginine in endothelial cells.
NO stimulates guanylate cyclase, increasing cyclic guanosine monophosphate (cGMP) production in target cells; which causes vasodilatation; and inhibits platelet adhesion and aggregation. The highlight of our comic today.
NO is rapidly destroyed by hemoglobin and thus functions as a paracrine hormone. (Accidental poisoning or overdose of a nitrate containing compound may cause methemoglobinemia)
Interesting facts: Intravenous infusion of an arginine analogue that blocks NO production leads to an immediate and substantial rise in blood pressure. This phenomenon suggests that NO is released continually and basally to regulate vascular tone.
Its effect on vascular tone is exploited in the treatment of angina. Nitroglycerin, taken sublingually, acts rapidly to cause vasodilation and decrease anginal pain. It can also be taken prophylactically for angina.
Related post: Anti-platelet drugs, receptor and their deficiencies mnemonic
545 notes
·
View notes
Text
—Es antinatural. ¿Tú alguna vez has visto a dos monos dándose por el culo? ¿O a dos perras comiéndose el coño?
—Lo que sí he visto es porno lésbico en tu portátil.
677 notes
·
View notes
Photo

Un profesor enseña un billete de $ 20.000 a sus alumnos y les dice: “¿A quién le gustaría tener este billete?” todos los alumnos levantan la mano.
Él arruga el billete y les pregunta de nuevo: “Ahora, ¿quién lo quiere?” Las manos suben de nuevo.
Él lanza el billete arrugado en el suelo, lo pisa y vuelve a preguntar: “¿Aún lo quieren? “
Los alumnos responden que sí y levantan la mano.
Entonces los miró y les dijo:
“Amigos míos, ustedes han aprendido una lección muy importante el día de hoy: Aunque he arrugado el billete, lo he tirado y lo he pisoteado ustedes quieren todavía el billete, porque su valor no ha cambiado, sigue con un valor de $ 20.000.
Muchas veces en la vida, te ofenden, personas te rechazan y los acontecimientos te sacuden. Sientes que ya no vales nada, pero TU VALOR no cambiará NUNCA para la gente que realmente te quiere. Incluso en los días en que estés en tu peor momento, TU VALOR SIGUE SIENDO LO MISMO”. Los alumnos aplaudieron emocionados…
No dudes nunca de tu valor… Siempre valdrás mucho para las personas que te quieren. NUNCA pero NUNCA valdrás menos, aunque así lo sientas.
13K notes
·
View notes

























